Three Wire Kidney Stone Retrievial Basket (for China Mainland Market)
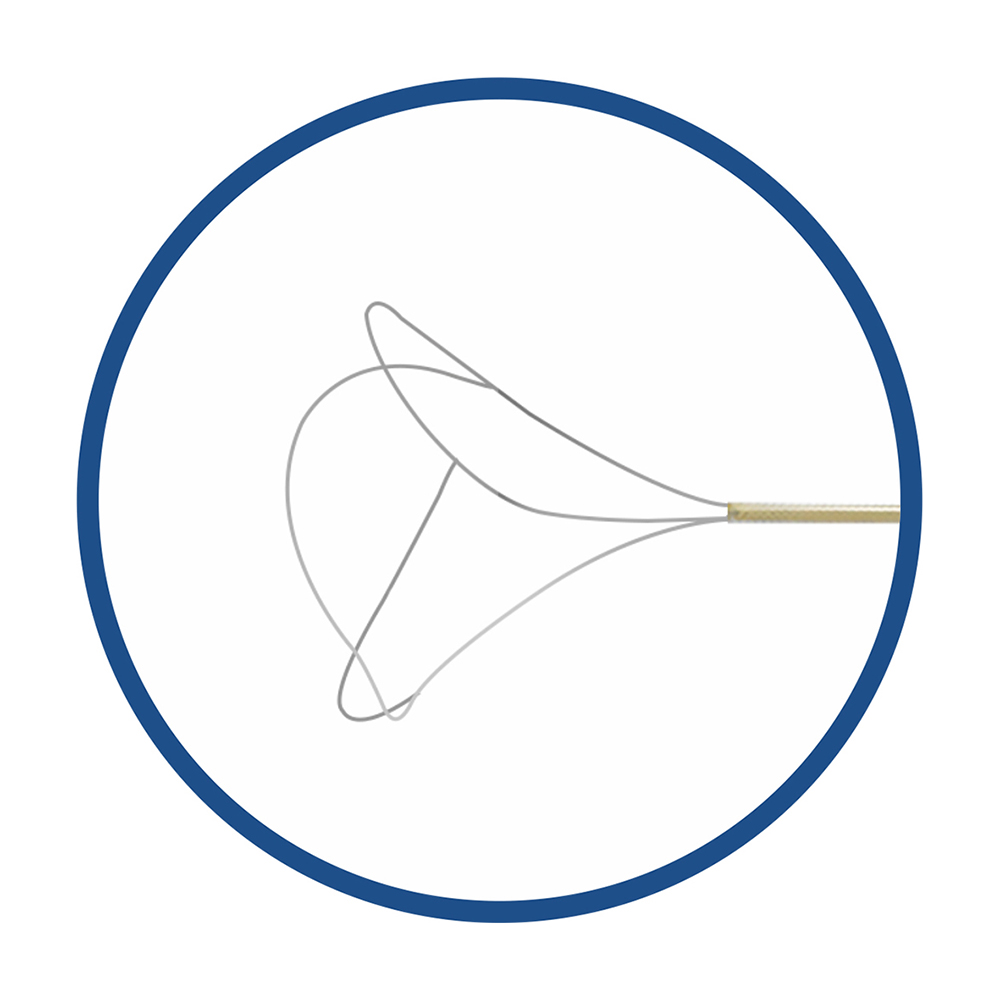
CE MDR FDA certified 3 -wire tipless basket is used to retrieval the stone from its lateral, 3-wire basket has a flat and wide basket opening, it is easily to capture the stone directly.
Intented use: disposable Kidney stone retrieval basket is used to to capture, reposition, and remove stones under the endoscope in the kidney, bladder, or ureter.
Working principle: its working part can be inserted into the natural orifice of the human body, get close to the stone, foreign body or tissue sample, the stone and foreign body can be captured by the mesh basket, and then the stone and foreign body can be firmly grabbed by the contraction of the mesh basket.
Environment of intented use: it is intended to be used in hospitals, clinics, etc., and should be operated and used by trained medical professionals. This product is for single use and is valid for three years from the date of sterilization. The product shall not be used after the expiration date of sterilization.
Applicable population: Patients with kidney stones and ureteral stones who need endoscopic lithotripsy, such as Retrograde intrarenal surgery (RIRS), percutaneous nephrolithotomy (PCNL), etc.
The disposable kidney stone retrieval basket is composed of mesh basket, guide rope, rope sheath, control handle and protective tube.
More