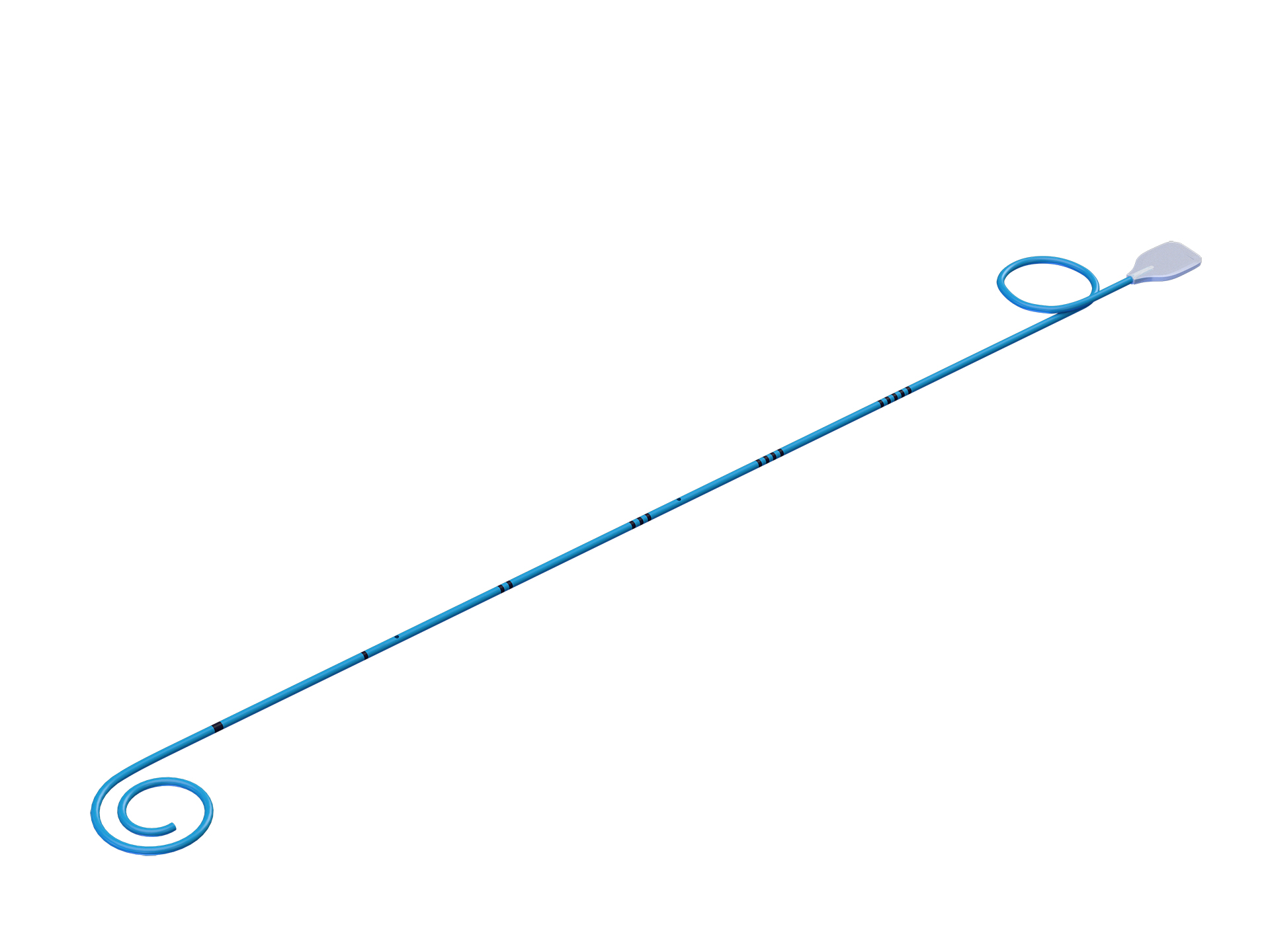
Anti-backflow Disposable Double J Ureteral Stent
This anti-backflow ureteral stent has a thin film in end of the loop that will be placed in the urinary bladder, this film will prevent the urine backflow to the ureter.
Working Scope: Under the guidance of medical instruments such as nephroscope, and ureteroscope, by placing ureteral stents in human ureters. It supports and drains the ureter after operation, helps the postoperative recovery, and prevents ureter blockage and stenosis.
Expected Usage Environment: This product is intended to be used in hospitals and clinics, and should be operated by trained medical professionals. This product is for single use and is valid for two years from the date of sterilization. Product over sterilization expiration date may not be used.
Applicable groups: It is suitable for ureteral support and support in patients with ureteral obstruction and stenosis after urinary surgery drain it.
Certification: US FDA, CE, MDR, ISO13485, FSC.
More