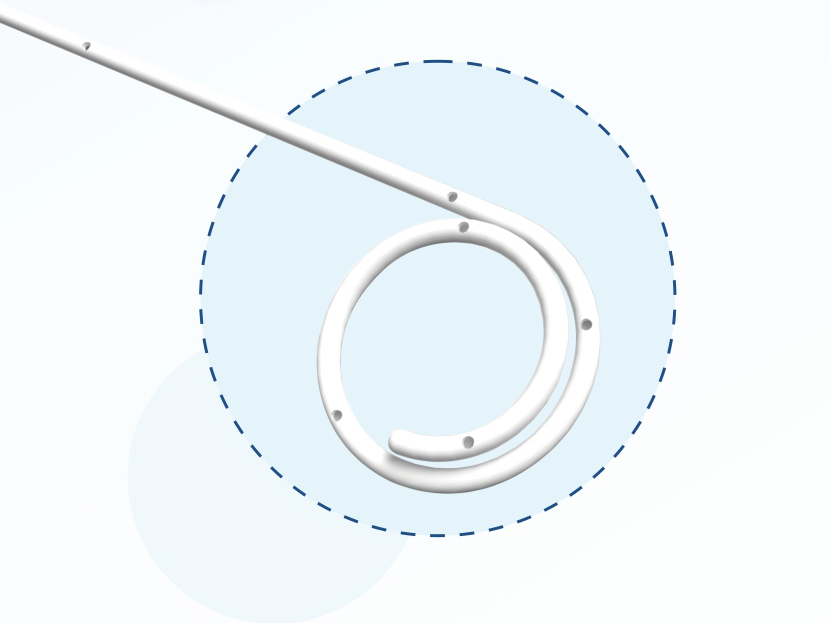
The flow rate of stent should be not less than 5ml/min;
Indications:
1) Stenting after ureteroscopy
2) Obstructing ureteral and/or renal stone
3) Pyonephrosis
4) Malignant ureteral obstruction
5) Ureteral stricture
Intended clinical benefits:
1) Easy insertion and removal,
2) Easy manipulation,
3) Resistance to encrustation and migration,
4) Lack of complications, biocompatibility, radio-opacity,
5) Biodurability,
6) Affordability (cost-effectiveness),
7) Tolerability and optimal flow characteristics.
This device is intended to be used for adult patients.
This product shall only operated by trained and qualified doctors.
1) Stent malposition
2) Inefficient drainage
3) Hematuria
4) Urinary tract infection
5) Stent encrustation and calcification
6) Stent fragmentation
7) Pain
1. DO read this instruction carefully before using, the product should only be operated by trained and qualified doctors.
2. DO inspect the product before using. It is FORBIDDEN to be used if there are abnormal conditions such as package breakage, dirt, moisture and product damage during the inspection;
3. Before use, please note that the working channel of the endoscope should be larger than the outer diameter of the ureteral stent, the inner diameter of the ureteral stent should be larger than the guide wire diameter, and the guide wire should be longer than the ureteral stent;
4. Be gentle when advancing or withdrawing the stent, pause the surgery if the moving is difficult and analyze the reasons to avoid potential medical negligence or device damage.
5. Record the time after placement, the stent tube should be reserved inside the body for less than 30 days. If it exceeds 30 days, there may be a risk of infection, urinary tract irritation, hematuria or pain, or difficulty in removal.
6. This is a single use device. Do not reuse, reprocess or resterilize. Reuse, reprocessing or resterilization may compromise structural integrity of the device and/or lead to device failure which, in turn, may result in patient injury, illness or even death. Reuse, reprocessing or re-sterilization may also create a risk of contamination of the device and patient infection or cross-infection from one patient to another. Contamination of the device may result in patient injury, illness or even death.
7. Do not change the structure of the product during use;
8. Patients should not engage in strenuous exercise during the use of this product and should notify their healthcare provider promptly if any discomfort occurs;
9. This product is sterilized using ethylene oxide and is for one-time use only. The validity period of sterilization is three years, it is FORBIDDEN to be used if the package is damaged or the product exceeds the validity period;
10. Products after use are disposed of in accordance with the regulations and procedures concerned with medical device waste.
11. The device is not to be used in a MRI environment.